Index
Main gases detected by gas sensors
| Gases | chemical formula | Color Smell | |
|---|---|---|---|
| Hydrogen | H2 | Transparent/Odorless | Used as fuel |
| Ammonia | NH3 | Transparent/Pungent odor | Used as fuel |
| Methane | CH4 | Transparent/Odorless | Main component of natural gas |
| Carbon monoxide | CO | Transparent/Odorless | Caused by incomplete combustion of fuel |
| Carbon dioxide | CO2 | Transparent/Odorless | Contribute to global warming |
| Sulfur dioxide | SO2 | Transparent/Pungent odor | Causes of acid rain |
| Nitric oxide | NO | Transparent/Odorless | Causes of photochemical smog |
| Dioxin | NO2 | Reddish brown/Pungent odor | Air pollutants |
| Ethylene | C2H4 | Transparent/Aromatic odor | plant hormones |
| Hydrogen sulfide | H2S | Transparent/Rotten odor | Bad breath ingredients |
| Acetaldehyde | CH3CHO | Transparent/Pungent odor | alcohol metabolites |
| Acetone | CH3COCH3 | Transparent/Pungent odor | Produced by the breakdown of body fat |
Gas sensors are used in a wide range of applications, including:
- Managing hazardous gases and helping prevent fires caused by flammable gases
- Monitoring odors and carbon dioxide to create a comfortable indoor environment
- Measuring environmental gases, including greenhouse gases
- Detecting gases released by living organisms
The gases shown in the table above are just a small example of what gas sensors can detect.
In our everyday surroundings, there are many different types of gases, including those emitted by industrial activities, released by animals and plants, and naturally present in the atmosphere.
What can gas sensors do?
Detecting odorless gases
Some odorless gases can present health or flammability hazards, making concentration monitoring necessary. Even if the concentration of such gases increases, humans are unable to perceive them. The ability to detect odorless gases is one of the key advantages of gas sensors.
Below are some examples of odorless gases that can be detected.
| Characteristics | |
|---|---|
| Carbon dioxide | Causes asphyxiation at high concentrations. |
| Carbon monoxide | Causes serious poisoning at high concentrations. |
| Nitric oxide | Harmful substance that causes photochemical smog |
| Hydrogen | Flammable gas |
| Methane | Flammable gas |
Detection of low-concentration gases that humans cannot perceive
One of the applications of gas sensors is the detection of gases at low concentrations that humans cannot perceive.
As shown in the table below, many gases with an odor can have harmful effects on the human body. Such hazardous gases should ideally be detected before people become aware of them by smell.
However, for some gases such as sulfur dioxide and nitrogen dioxide, the human olfaction can detect them at lower concentrations than gas sensors. Accordingly, gas sensor development aims to achieve sensitivity exceeding that of human olfaction for these gases.
| The minimum concentration at which humans can perceive an odor(detection threshold) | |
|---|---|
| Ammonia | 1.5ppm |
| Sulfur dioxide | 0.87ppm |
| Dioxin | 0.12ppm |
| Hydrogen sulfide | 0.00041ppm |
| Acetaldehyde | 0.0015ppm |
Detection of toxic gases
The detection of toxic gases is one of the key roles of gas sensors. Smelling toxic gases is hazardous to human health, which is why gas sensors are essential.
Gas sensors detect the presence of toxic gases such as ammonia, carbon monoxide, and hydrogen sulfide and help reduce exposure risks.
Detection of flammable gases
Early detection of leaks of flammable gases such as methane and hydrogen is important for preventing fire accidents. Gas sensors play a vital role in monitoring gas leaks.
Gas monitoring in unmanned environments
Gas sensors also enable air-quality monitoring in unmanned environments.
Examples include measuring environments with extremely low oxygen levels where humans cannot enter, detecting leaks of flammable gases in automated (unmanned) factories, and monitoring air conditions inside greenhouses around the clock. Gas sensors are indispensable for monitoring unmanned or restricted-access areas.
Human olfaction vs. Gas sensors
So far, we have introduced the main roles of gas sensors. To summarize the basic concepts of gas sensors, the advantages and disadvantages of detection by the human sense of smell and by gas sensors are outlined below. Gas sensors can detect substances that cannot be perceived by human olfaction and gases that are dangerous to smell. In this way, gas sensors play a role in supporting human olfaction.
| Human olfaction | Gas sensors | |
|---|---|---|
| Detecting odorless gases | Undetectable | Detectable |
| Detection of toxic gases | Hazardous to smell | Detectable |
| Continuous monitoring | Sensitivity decreases over time | 24/7 operation possible |
| Performance stability | Affected by physical condition | High performance stability |
| Odor discrimination | Odor discrimination | No odor discrimination |
Types of Gas Sensors
Semiconductor gas sensor
This type of sensor uses semiconductors such as tin oxide (SnO₂) as the gas-sensing material.
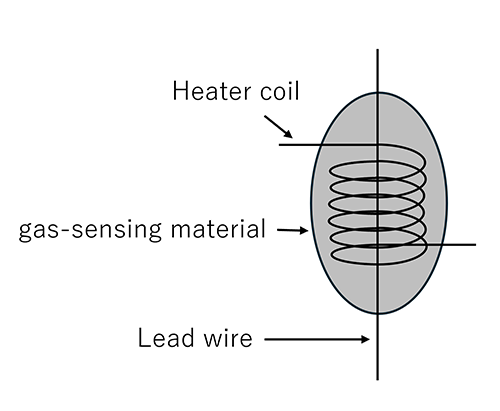
Semiconductor gas sensors incorporate a heater to heat the gas-sensing material.
When gas molecules are adsorbed onto the surface of the heated gas-sensing material, its electrical resistance changes. Gas detection is achieved by monitoring this change in resistance.
By optimizing the semiconductor materials and catalysts used as the gas-sensing material, it is possible to detect specific gases depending on the application.
Catalytic combustion gas sensor
This sensor consists of a platinum coil covered with an inorganic support material containing a catalyst.
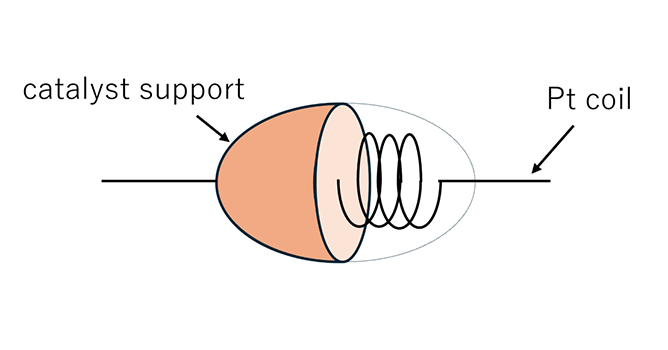
When the heated catalyst undergoes a combustion reaction with a flammable gas, the resulting temperature rise changes the resistance of the platinum coil. Gas detection is achieved by monitoring this change in resistance.
Catalytic combustion sensors use a pair of elements: a sensing element and a compensating element. A typical example of their application is hydrogen
(H2)sensing.
Electrochemical gas sensor (fuel cell type)
This sensor has a structure similar to a battery, in which an electrolyte solution is filled between a sensing electrode and a counter electrode.
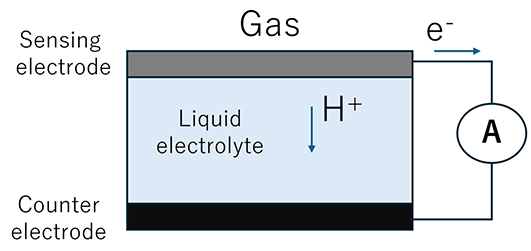
When a gas comes into contact with the sensing electrode, an oxidation reaction occurs, generating hydrogen ions and electrons. The electrons flow through an external circuit toward the counter electrode, and the gas is detected by measuring the resulting current.
Electrochemical sensors are mainly used for carbon monoxide (CO) detection.
Infrared (IR) absorption gas sensor
This sensor detects gas by utilizing infrared absorption by gas molecules and measuring the decrease in infrared radiation intensity emitted from a light source.
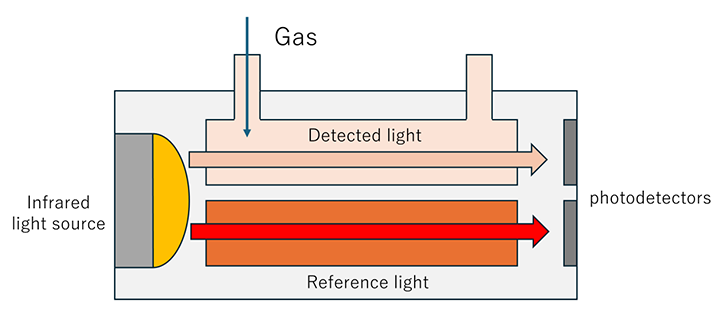
An infrared light source is located at one end of the cell (optical path), and a photodetector is positioned at the opposite end. Gas is passed through the cell, and the sensor detects the gas by comparing the infrared radiation intensity in the gas-filled cell with that in a gas-free reference cell.
This method is commonly used in sensors such as CO₂ sensors.
Solid-state electrolyte gas sensor
Unlike electrochemical sensors, which use an electrolyte solution, solid-state electrolyte gas sensors use a solid electrolyte such as ceramics.
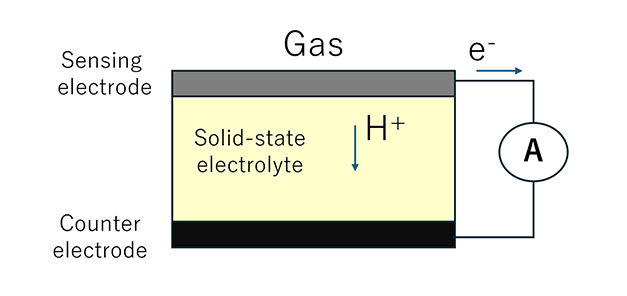
The sensor structure, consisting of a sensing electrode, an electrolyte, and a counter electrode is the same as that of electrochemical sensors. Because no liquid electrolyte is used, there is no risk of leakage or degradation due to drying, resulting in stable performance.
Detection concentration ranges by gas sensor type
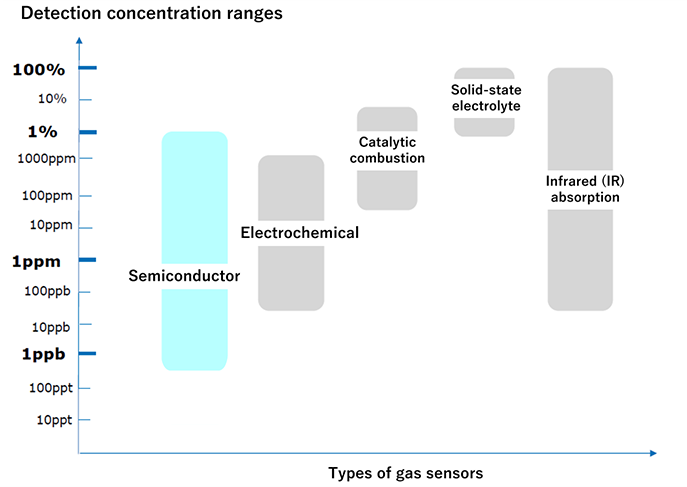
The detectable concentration range of gas sensors varies depending on the sensor type. Therefore, it is necessary to select the appropriate sensor type based on the target gas and the concentration range to be measured.
Nissha FIS is a specialized manufacturer of gas sensors
Nissha FIS provides gas sensors for wide range of target gases, with a primary focus on semiconductor-type gas sensors. Visit our website for product information and lineup details.
Products information web site >
[Products topics]
Refrigerant sensors
Designed for detecting refrigerant gas leaks, including fluorocarbons.
Click here to product information >
Hydrogen detectors
Designed for detecting hydrogen leaks in hydrogen fuel cells and other hydrogen applications.
*The hydrogen detector is a product offered by Nissha Co., Ltd.


